What's new
Patients in a clinical trial get treated faster thanks to digital pathology
The paper J. Hudecek et al.: Application of a risk-management framework for integration of stromal Tumor Infiltrating Lymphocytes in clinical trials published today in Nature Breast Cancer describes how Slide Score was used in the TONIC clinical trial to minimize potential risks.
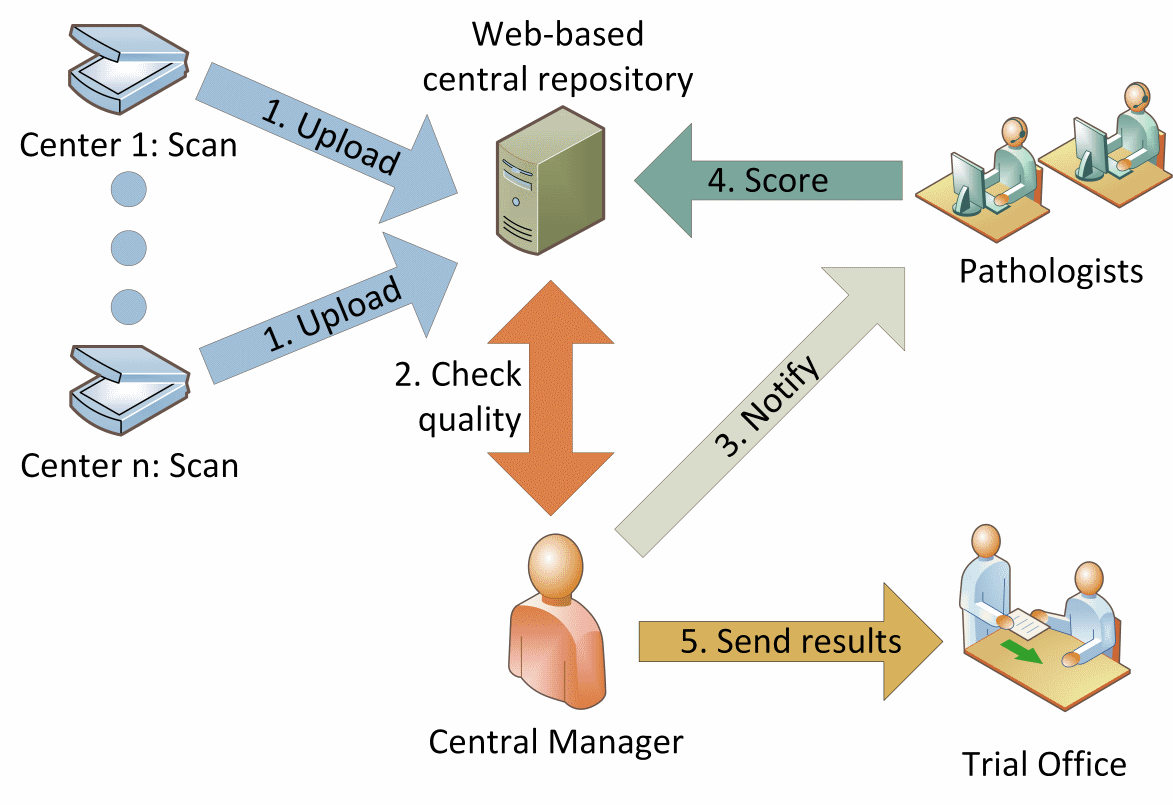
A slide from a patient sample was scanned and then uploaded to Slide Score using the anonymizing Simple Uploader. Study coordinator notified four expert international pathologists about availability of new slide and they assessed it for percentage of tumor infiltrating lymphocytes (TIL). The consensus score was used to assign patients to one of two treatment arms - high or low TIL.
Using Slide Score for pathology support in a clinical trial simplified distributing the slides and collecting the assessments from pathologists in three different institutions. They were able to view the slides at home or on-the-go which contributed to very short times needed to get a consensus score. Telepathology and great teamwork helped reduce the time patients had to wait before continuing treatment - the median time between biopsy (taking a sample) and assignment to a treatment arm was reduced from usual 10 days to just over 2 days.
Slide Score can effectively mitigate some of the risk factors in biomarker-driven clinical trials.
Contact us for help with your clinical trial!